Struvite Scale and Its Chemistry
- Oct 6, 2023
- 22 min read
Updated: Jul 13, 2025
For the last several years I have been doing a lot of field testing to determine the potential for struvite scale to form in anaerobic digesters and dewatering equipment (primarily centrifuges). In this post I will be focusing on struvite scale that forms in centrifuge centrate lines, with the "centrate" or "filtrate" line from dewatering equipment being one of the most common locations where struvite forms in wastewater plants with anaerobic digestion. For the wastewater treatment plant operator, if you have a struvite problem developing you typically won't know until it is too late. That's becuase you often have no direct indication that your centrate/filtrate line is begining to narrow as scale forms on the inside of the pipe wall and the flow gradually, but increasingly, becomes restricted.
In Figure 1 you can see a large chunk of struvite scale removed from an 8-inch centrifuge centrate gravity flow line. The diameter of this line reduced over time causing the centrate to finally back up into the concrete containment area underneath the centrifuge. Too often, it is only when this happens that you know you have a struvite scale problem! And when this does happen, the entire sludge dewatering operation may need to be shut down for several days at a time, while plant maintenance staff and/or a subcontractor removes the scale, causing a major disruption to plant operations. Once struvite scale has formed in the centrate line the task of scale removal can be enormous, requiring acidification (pH ≤4.5) to dissolve the scale combined with high pressure line cleaning, sometimes requiring the removal of multiple pipe segments to gain a sufficient level of access.
Figure 1

Fair Warning: To study struvite, to understand it, and to perform chemical arithmetic, we will need to think like a chemist (as best we can!), adding this viewpoint to how we normally think as a wastewater treatment plant operator or as an engineer. I say this to you because I am an operator, not a chemist, and chemistry has become, almost by necessity, my most favorite area of study over the last few years. If you, like me, find yourself needing to increase your knowledge of chemistry, I have provided a list of recommended textbooks at the end of this post. In the meantime, as we proceed to talk about struvite, I am going to provide chemistry details, definitions, and explanations wherever I think they might be helpful. To a chemist this may be boringly simplistic but, perhaps and hopefully, this won't be the case for the operator or the engineer.
What Is Struvite?
Struvite scale is a crystalline substance comprised of Magnesium, Ammonium, and Phosphate (MAP) and forms in water as shown by the chemical equation (Equation 1) below. This seemingly simple equation is referred to as a synthesis or combination reaction (A + B ―> AB) and it is one of the most important activities of chemistry. This is also a precipitation reaction because the oppositely charged ions on the left side of the equation attract one another so strongly that they form an insoluble solid (s) as shown on the right side of the chemical equation.
Equation 1
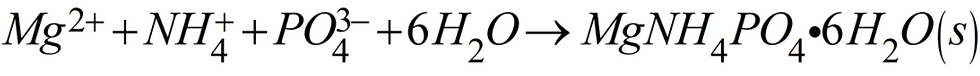
The Chemistry of Struvite: An Introduction
Equation 1 tells us that one magnesium (Mg) ion combines with one ammonium (NH4) ion, one phosphate (PO4) ion, and six molecules of water (6H2O) to form struvite when the pH (>7), conductivity, and wastewater temperature are at the required levels. Another way to state what Equation 1 is telling us is that 1 mole of magnesium + 1 mole of ammonium + 1 mole of phosphate + 6 moles of water combine to form 1 mole of struvite.
Mole Definition: The chemistry term "mole," often abbreviated "mol," is a unit of measure used to count atoms and molecules in a sample. More specifically, a mol is "the number equal to the number of carbon atoms in exactly 12 grams (g) of pure carbon-12. This number (6.022 x 10^23) is called Avogadro's number. Most important is that a mole is a unit that allows us to easily count incredibly huge numbers of infinitesimally small objects.
How do we plan to use moles? I used the phrase "chemical arithmetic" earlier and this math is all about the quantitative relationships between the ions and compounds that combine to form struvite (or any compound), with chemical formulas and equations guiding our mathematical operations. This is way too wordy, I know, so let's get started by looking at, and doing, some of the arithmetic.
We will begin with the element magnesium. (In Equation 1, mangesium is an ion as indicated by its charge of +2.) From a periodic table of the elements, a subset of which is shown in Figure 2, we can see that magnesium (Mg) has an atomic mass of 24.31. With this information we can easily and directly determine the molar mass of magnesium.
Molar Mass Definition: The molar mass is the mass, in grams, of a substance that is numerically equal to that substance's atomic mass.
So the molar mass of magnesium is 24.31 grams.
Figure 2
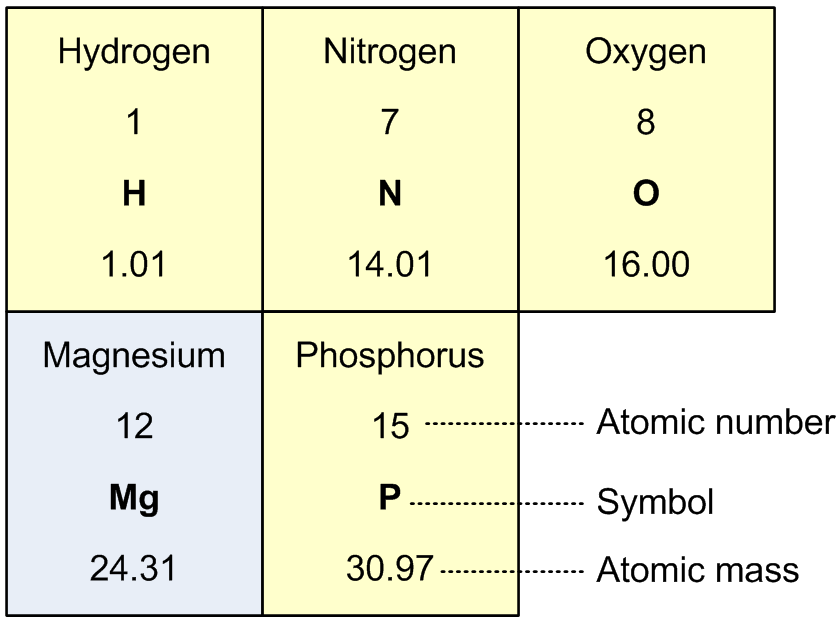
Next up is the ammonium ion, NH4. Taking guidance from Equation 1 and information from Figure 2 we can calculate the molar mass of ammonium as [(1 x 14.01) + (4 x 1.01)] = 18.05 g. The molar mass of the phosphate ion, (PO4), is [(1 x 30.97) + (4 x 16.00)] = 94.97 g. Finally, for the six molecules of water, 6H2O, we have a molar mass of 6 x [(2 x 1.01) + (1 x 16.00)] = 108.12 g. These calculations are summarized in different ways in Tables 1 and 2.
Note that in Table 2 it would probably be more "appropriate" to change the columm heading with "Substance" to "Element" but I really want you to see how I set up my Excel spreadsheets. Everyone has their own approach and all I can do is show you mine, hoping I am not abusing terms or adding confusion where little should exist.
Table 1
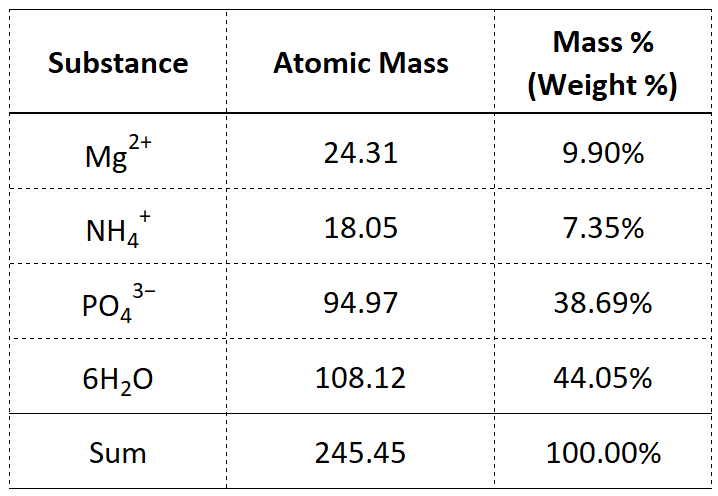
Table 2

The composition of struvite by mass percent, derived from the chemical equation itself (Equation 1), and the calculated atomic mass values listed in Table 1, is shown in the pie chart (Figure 3).
Figure 3

I had the struvite scale (sometimes referred to as a phosphate mineral crystal) shown in the photograph (Figure 1) analyzed and the results are summarized in Table 3 below. As you can see from the percentage values, what the chemical equation (Equation 1 and Table 1) predict is different from what actually happens in the complex environment of an anaerobic digester where so many competing reactions are taking place. There are several things worth pointing out in Table 3. For example, though "pure" struvite consists of 9.90% magnesium on a mass basis, (as shown in Table 1 and the pie chart in Figure 3), this sample is 31.3% magnesium. Likewise, though phosphate makes up 38.69% of the mass in struvite scale, this sample contained 49.6%, perhaps lending justification to struvite being described as a phosphate mineral crystal. The 16.0% loss on ignition value measured the mass of organic matter bound into the struvite sample.
Table 3

Regarding the struvite scale analysis in Table 3, my final comment is about the iron oxide value of 0.2 percent. Though this post is going to focus on struvite scale, another potential scale of concern is vivianite. This concern is necessary when an iron salt (e.g., ferric chloride, ferrous chloride) is being added to the anaerobic digester for hydrogen sulfide reduction, a fairly common practice. With a sufficient iron concentration in the digester, in addition to struvite scale, the operator may need to worry about vivianite scale formation. You will see the chemical formula for vivianite in different ways as shown in Equation 2. This equation tells us vivianite is comprised of ferrous iron (Iron II) and phosphate in eight waters of hydration. Ammonium is not a component of vivianite scale and iron is not a component of struvite scale.
Equation 2
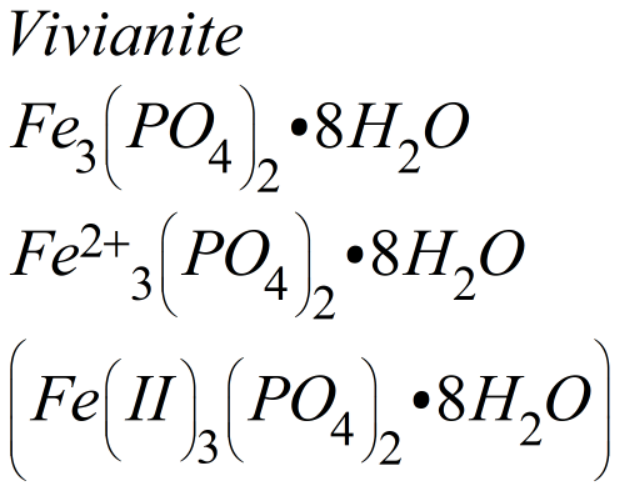
Struvite Scale Formation Requires Anaerobic Digestion
In my experience, if you do not have anaerobic digesters you do not have to worry about struvite scale. This view is suppported in the research literature as well. The process flow diagram in Figure 4 shows laboratory results for the influent, effluent, and centrifuge centrate in a wastewater plant with anaerobic digestion. You can see there is no struvite scale formation in the influent or the secondary clarifier effluent. Struvite scale only forms as a result of favorable conditions that are created in the unique environment of an anaerobic digester. The anaerobic digestion process, by means of volatile solids reduction/destruction, is critical to producing soluble concentrations of magensium, ammonia, and phosphate (MAP) high enough to crystalize. You won't find struvite scale to be a problem in the rest of the wastewater treatment plant (influent, screenings, primary clarification, activated sludge, secondary clarification, disinfection, etc.).
Figure 4

In Figure 5 below, you can see the solids processing part of another wastewater treatment plant, a different plant from the one represented in the flow diagram above (Figure 4). Notice in particular, in Figure 5, that the sludge feed to the gravity belt thickener (GBT) and the filtrate from the GBT both have negative log of saturation index values (described later in this post). In both cases no struvite is formed. Also notice that the plant reuse water (plant effluent) shows no potential for struvite scale to form.
It is only after anaerobic digestion and the breakdown of microbiological cells that sufficient concentrations of MAP are released into the digesting sludge. The sludge flow out of the digester (digestate) has a positive saturation index and this indicates that struvite scale can form in the digester, sludge recirculation lines, sludge pumps, heat exchangers, and dewatering equipment. At this wastewater plant, the digestate is transferred to a digested sludge holding tank providing additional retention time that contributes to more cell destruction, increasing the saturation index in the centrifuge feed sludge and centrate. (If you click on any graphic it should open up as a larger image on its own page allowing you to see more detail.)
Figure 5

Struvite Data Summary and Discussion
I want to share some of the data I've collected doing struvite testing. This data is from 23 samples collected at three wastewater plants across the United States. The software I use to calculate struvite formation is a simple program called, appropriately enough, Struvite Tool. The program is an Excel add-in that requires seven inputs to calculate the potential for struvite scale to form as portrayed in Figure 6. Further down in this post you can find more information about this software program, including a link to where you can order it.
If You Want to Be Proactive (And I think you should want to be!): The Struvite Tool is very easy to use and inexpensive (US$ 75) to buy and well worth considering if you have anaerobic digestion. If you are able to measure the seven parameters shown in Figure 6, entering this data into the Struvite Tool will allow you to monitor the struvite scale potential in any stream of interest to you.
Figure 6
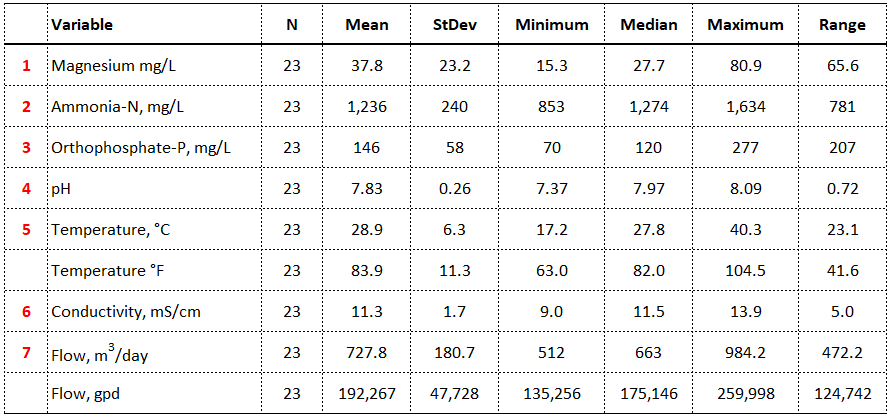
A data summary of the required inputs from the 23 centrate samples (collected over a two-year period) is provided in Table 4. The numbers in red indicate the inputs and the unit of measure. The two unnumbered rows have been converted to U.S. Customary units for those of us, like me, who do not regularly use the metric system.
Table 4
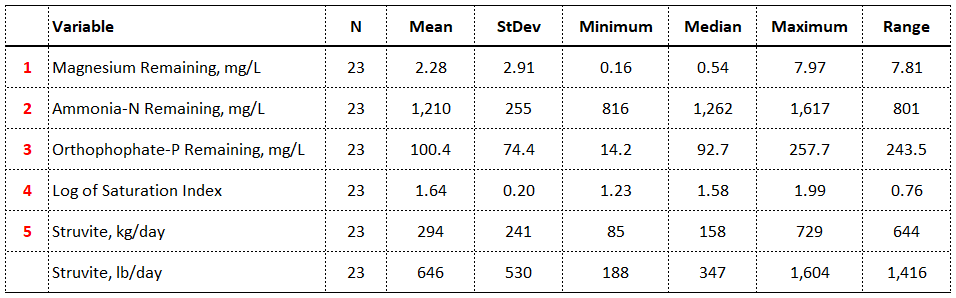
Once the seven inputs to the Struvite Tool are entered into the program, five results are generated as shown in Table 5. Struvite scale has the potential to form whenever the log of the saturation index is >0. When the saturation index (SI) is <0, the sample being analyzed is undersaturated. When the SI is >0, the sample is oversaturated.
Table 5
Something I find interesting from Tables 4 and 5 is the remaining concentration of MAP in solution after the struvite scale has formed. In Table 6, below, you can see that 94% of the available magnesium (on average) goes into the development of struvite. Next is the 31.5% consumption of orthophosphate. Last, unsurprisingly, because its concentration is so high to begin with (bar graph in Figure 7), we get just a 2.1% utilization (consumption or combining) rate for ammonia.
Table 6

Figure 7

Magnesium Is the Limiting Reactant
Magnesium will be the ion that limits how much struvite can form. There will always be an excess of both ammonia nitrogen and orthophosphate in the anaerobic digester that has been released from cell breakdown. Therefore, no matter how much struvite scale is formed, the remaining concentration of unbound nitrogen and phosphate will be very high compared to the residual magnesium concentration. In the next section I'll show how the limiting reactant is determined through stoichiometry.
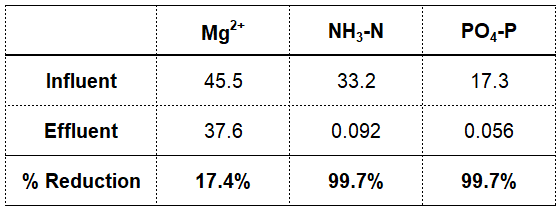
If we look at the process flow diagram in Figure 8 we can see the concentrations of magnesium, ammonia, and phosphate throughout this particular wastewater plant. The removal rates for ammonia and phosphate are outstanding at 99.7% for both parameters as shown in Table 7. This is great for effluent quality but it's not so good for providing high concentrations for two of the three key ions that combine to form struvite in the anaerobic digestion process.
So what about magnesium? Since just 17.4% of magnesium is removed with the sludge going to digestion that means 82.6% leaves with the effluent. That's a good thing when it comes to reducing struvite production because the magnesium ion is one over which the treatment plant operator has no direct control. There is no process control strategy or unit process that can effect the removal of magnesium. What this means is that the only control point an operator has to reduce struvite is to precipitate orthophosphate before it can become available to participate in struvite formation. For the time being this is just something we can think about.
Figure 8

Table 7
Stoichiometry: A Method to Determine Limiting and Excess Reactants
Stoichiometry Definition: Stoichiometry (pronounced "stoy-key-om-e-tree") is the calculation of the quantities of reactants and products involved in a chemical reaction. It is based on the chemical equation and on the relationship between mass and moles. (Ebbing and Gammon, 88)
We are back to our chemical arithmetic to determine the limiting reactant and those reactants that will be in excess of what is needed to form struvite. In the pair of identical equations below (Equation 3) I am calculating, and showing how terms and units cancel, the mass of magnesium that is available in the centrate that can contribute to the formation of struvite scale. Equation 3 tells us there 27,511 grams of magnesium available to combine with ammonium and phosphate to form struvite. Keep in mind that I am using the average magnesium concentration (37.8 mg/L) from the 23 centrifuge centrate samples along with the average centrate flow rate of 727.8 cubic meters per day. I also know the average struvite scale formed from the 23 samples was 294 kilograms (kg). All of this information is listed in Tables 4, 5 and 6, above.
Equation 3

If I let Sv represent the mass of struvite formed, based on the mass of magnesium available as calculated in Equation 3, the chemical math shown in Equation 4 tells us 277,769 grams (g), or 278 kg of struvite will form, consuming, in theory, all of the available magnesium. This calculated result of 278 kg is just 5.4% less than the actual value of 294 kg so our math looks good.
Equation 4

For reference, please recall that the 24.31 g molar mass value for magnesium and the 245.45 g molar mass value for struvite used in Equation 4 came from Table 2, which showed one way to calculate the molar mass. In Table 8 I take a somewhat different approach to derive molar mass values. It all has to do with how we each set up our spreadsheets and I know my "logic" is likely to be very different from your logic.
Let's also recall that "molar mass" is the quantity in grams that equals the atomic mass (many people use atomic weight) of that element, having used a periodic table to find the atomic mass (weight). From this we know now that 24.31 grams of magnesium contains 6.022 x 10^23 atoms of magnesium. In the case of ammonium, we know that 18.05 grams has 6.022 x 10^23 atoms of N (1 mole) and 4 x 6.022 x 10^23 atoms of hydrogen (4 moles). Clear as mud, right?
Table 8

There is another way to determine which of the three MAP reactants will be limited, thereby establishing the amount of struvite that can form. We can do this by comparing the amount of ammonium needed to combine with the available magnesium. To begin, we need to calculate the mass of ammonium available and this is shown in Equation 5 which calculates that 1,158,658 g of ammonium can participate in struvite formation. The careful reader might notice that Equation 5 has an ammonium (NH4) concentration of 1,592 mg/L which is quite different from the ammonia-N (NH3-N) concentration of 1,236 mg/L shown in Table 4, Table 6, and Figure 7. I will be explaining this critical difference further down in this post, in a section titled "Which Chemical Form Should I Be Using?" For now, accept the validity of using a concentration of 1,592 mg/L for ammonium and a concentration of 448 mg/L for phosphate in Equation 7.
Equation 5

Now we need to calculate the amount of ammonium needed to combine with all of the available magnesium and this is shown in Equation 6. From Equation 6 we can see that only 20,427 g of ammonium, from the 1,158,658 g available, are required, leaving an excess of 1,138,231 g of ammonium in solution after struvite has formed. Clearly, not only is ammonium not limited, in comparison to the limited quantity of magnesium the ammonium is nearly limitless!
Equation 6

Finally, we can repeat the approach taken with Equations 5 and 6 for phosphate. In Equation 7 the amount of phosphate available to form struvite is calculated to be 326,054 g. Note the 448 mg/L phosphate concentration used in Equation 7.
Equation 7
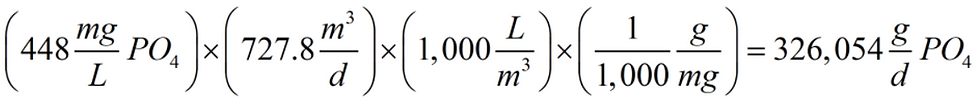
From Equation 8 we can see that, given the 27,511 g of magnesium available, we need to use 107,475 g of phosphate to fully combine to form struvite scale, leaving 218,579 g as excess phosphate reactant to participate in other reactions, biological for example, as the centrate becomes a recycle stream returned to the head of the wastewater plant.
Equation 8

We Are Done With Chemical Arithmetic!
Linear Regression Analysis
Three simple linear regression results from Minitab are presented below. In each analysis, one of the MAP variables is used to predict the formation of struvite. You can look at each of the fitted line plots to get a feel for how each MAP variable influences struvite. But should also look at the R-sq value in each model's summary. R-sq is the correlation coefficient with a range of 0 to 100 percent. The higher the R-sq number, the better the model fits the data indicating the strength of the linear relationship.
In the first analysis, shown in Figure 9, we want to know how well the moles of magnesium predict the kilograms of struvite formed. The R-sq value is 99.85%, indicating that moles of magnesium is an excellent predictor of how much struvite scale will form. And we learned the same thing about magnesium from the chemical arithmetic done in Equation 4.
As you will see in Figures 10 and 11, neither the ammonium nor the phosphate ions come close in predictive ability compared to the magnesium ion. The R-sq value for struvite vs moles of ammonium is 17.46% (Figure 10). And for moles of phosphate the R-sq value is a mere 0.21 percent (Figure 11).
Start of Regression Results
Figure 9
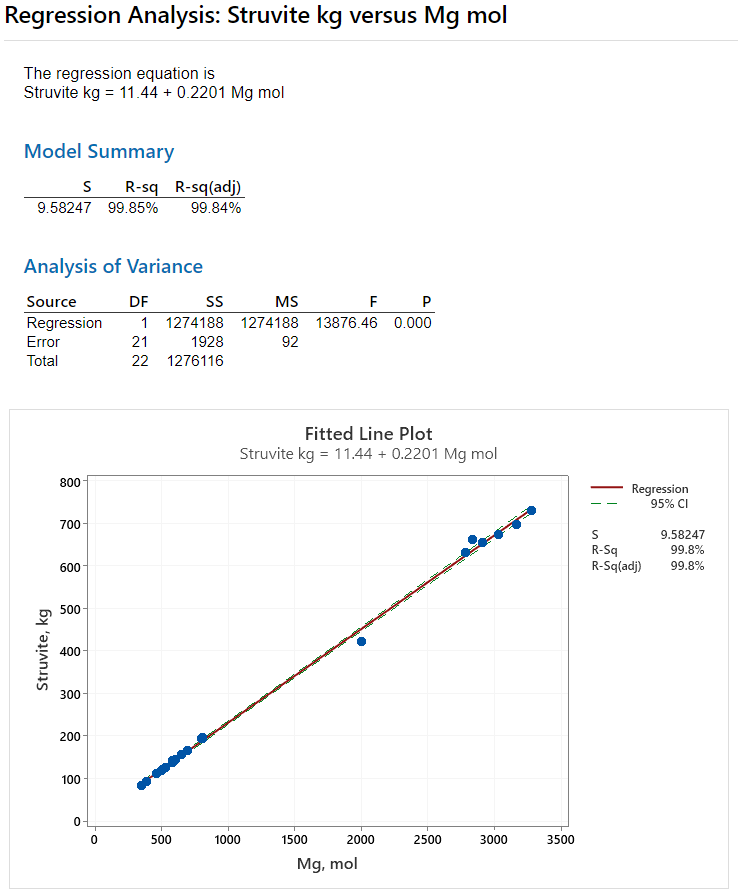
Figure 10
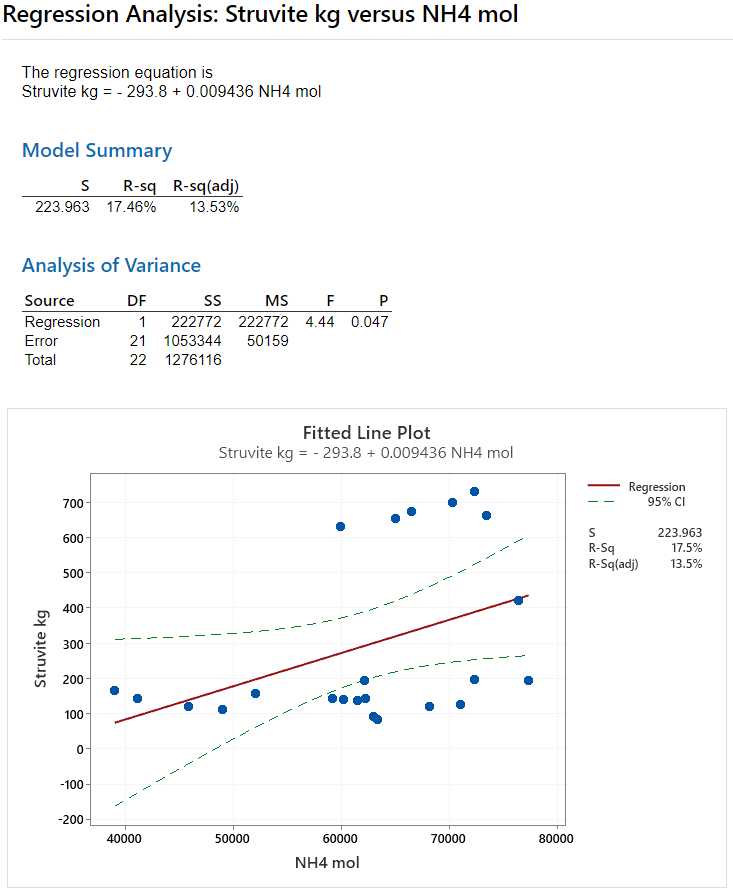
Figure 11

End of Regression Results
Which Chemical Form Should I Be Using?
In Tables 4 and 6 and the graph in Figure 7 above, I show the average concentrations of the MAP reactants which I want to list here.
Magnesium: 37.8 mg/L as Mg2+
Ammonia-N: 1,236 mg/L as NH3-N
Orthophosphate-P: 146 mg/L as PO4-P
Now let's recall the chemical equation for struvite (Equation 1, above, which I won't renumber here).
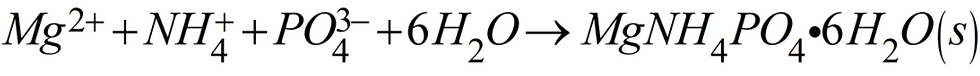
From this equation we can see that struvite is formed from magnesium ion (Mg2+). But things change now because struvite is formed from ammonium (NH4), not ammonia nitrogen (NH3-N), and from orthophosphate (PO4) not orthophosphate-P (PO4-P). This may seem trivial but it actually makes an enormous difference when doing chemical math. I think this can be confusing because the use of NH4 means you want the mass of both the nitrogen and the hydrogen whereas with NH3-N you just want the mass of the N after the dash. Likewise with phosphate, the use of PO4 means you want the mass of both the phosphorus and the oxygen whereas with PO4-P you just want the mass of the P after the dash.
Let's now list the concentrations of MAP using the "correct" units of measure required to form struvite.
Magnesium: 37.8 mg/L as Mg2+ (just as before)
Ammonium: 1,592 mg/L as NH4
Orthophosphate: 448 mg/L as PO4
Again, I find all of this to be so confusing if you are not very careful when doing the lab work so I want to spend some time going over these differences in the units of measure. I know if you are a chemist this all might seem a little ridiculous and unnecessary. But I'm a wastewater treatment plant operator who has become more and more involved with the chemistry of wastewater over the years and it has taken me a long time to become comfortable going back and forth between units while I continue to see people around makes these mistakes.
Keep Your Units of Measurment Straight!
It is critical that lab values we generate and report have the correct units and this is an area where it is so easy to make mistakes. Everything I am about to discuss here involves my use of Hach TNT (Test-iN-a-Tube) reagents using a Hach spectrophotometer (DR1900 or DR3900) and Hach probes to measure pH and conductivity mated to one of Hach's HQd handheld meters.
To do struvite testing we need to measure magnesium, ammonia, and phosphate (MAP) concentrations. Every treatment plant I go to has different wastewater characteristics and every sample location has different chemical concentrations. That means I spend my first day of testing just doing "range-finding" by selecting from the reagents and ranges listed in Table 9. (Total phosphorus requires heating using a digital reactor.)
Table 9
When measuring magnesium, for example, we get a straightforward result in the unit of Mg2+ from the spectrophotometer. And this magensium value can be used directly to calculate struvite potential. But when it comes to ammonia and phosphorus (or phosphate or orthophosphate) things can become very confusing!
Ammonia Testing
I've been in the field collecting samples, doing dilutions (which only adds to the confusion and chaos), and running tests, side-by-side with others. One person might be measuring pH, temperature, and conductivity, all easy tasks because you are using a probe. Another person is measuring ammonia concentrations and perhaps I am running the magnesium and phosphate tests. At the end of the day I get emailed ammonia values without any units. So I have to ask if the values are as ammonia (NH3) or ammonia-nitrogen (NH3-N)? And the person can't remember how the spectrophotometer was set up, which unit was used for the measurements. That means we have to wait until the next day to go back to the plant and go through the stored data on the spectrophotometer to determine the units of measurement. That's a good day. But it has also happened where the spectrophotometer has been boxed up and shipped to another site and I can't use the data until I know the unit of measurement. That makes for NOT such a good day!
The spectrophotometers I use can measure ammonia in either the unit of NH3-N (ammonia nitrogen) or NH3 (ammonia). This is a user-selected option. And really, it doesn't matter which unit you use as long as you record and report the value correctly and perform any required unit conversions. The spectrophotometer will not give you a result in the unit of NH4, ammonium, which is the ion that contributes to the formation of struvite. When I enter the ammonia concentration into the Struvite Tool, the unit must be as ammonia nitrogen, NH3-N. If my spectrophotometer is set up to measure ammonia (NH3) and I use this value in the software program I will be using the wrong value, a higher value, which I'll explain.
Look at Table 10 below to see what I'm talking about. Ammonia-nitrogen gives you just the N or nitrogen value. Ammonia (NH3) has a formula mass 1.216 times greater than nitrogen alone because hydrogen is included. And ammonium has a formula mass 1.288 times greater than nitrogen alone because you are now counting the mass contribution from four additional hydrogen atoms. I often set these tables up in my spreadsheets so I have all the units available at a glance. For many purposes we don't care about the hydrogen. But when we do chemical arithmetic the formula mass becomes critical and we will have to convert between units. This type of table makes doing so very easy.
Table 10

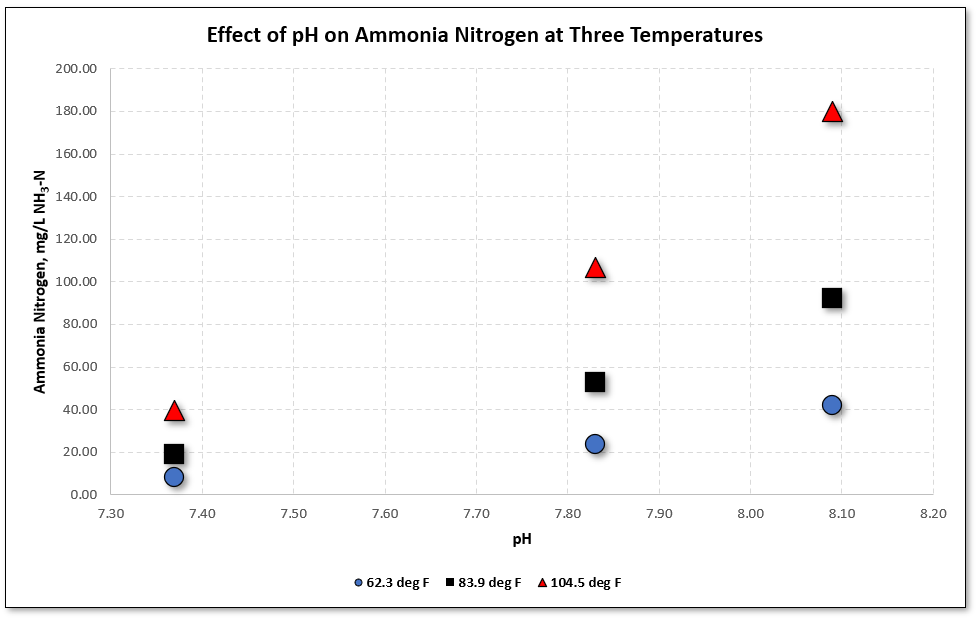
While we are talking about ammonia and nitrogen I should mention that both pH and temperature influence the concentration of ammonia nitrogen as shown Figure 12. If you want to evaluate this for yourself you can download an Excel spreadsheet (Unionized Ammonia Calculator) provided below Figure 12.
Figure 12
Phosphorus Testing
Now we need to talk about phosphorus where things don't get any better than they were with ammonia. Phosphorus can be measured on a Hach spectrophotometer using any of the three units shown in the Excel table, Table 11, below (very similar to the setup used for ammonia in Table 10). I never use the phosphorus pentoxide unit though if you work with soils, for example, this might be exactly what you want. I use only the orthophosphate as P or as orthophosphate with both P and O (oxygen). And I can't tell you how many times I have messed up by not keeping track of how my spectrophotometer was set up. You must pay close attention to the unit you are using when measuring, when recording, when reporting, and when calculating!
Table 11
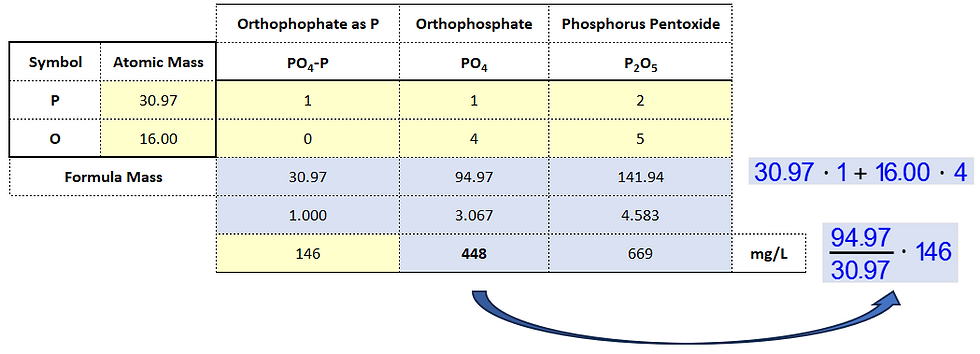
The struvite software wants the phosphate value to be entered as P. But struvite forms from the orthophosphate ion, PO4, which has a formula mass more than three times greater than the mass of P alone. So you need to pay attention and it's only because of my own pain and suffering that I keep bringing this to your attention in the hope I can save you some trouble.
Note that the struvite calculation uses the reactive phosphorus (orthophosphate) test. If you wanted to know the total phosphorus concentration that would require a digital reactor to heat the reagent vial in the reactor for 1 hour at 100 °C (212 °F) or for 30 minutes at 120 °C (248 °F).
Recommendation: There is an excellent, short, 4-page PDF file from Hach that I highly recommend you download and read with the title as shown in the graphic below. The download link follows immediately after. Good, very informative reading!

Conductivity Testing
I have seldom needed to measure the conductivity of a wastewater sample. I think this measurement is perhaps more meaningful to people working with cooling towers and boiler feedwater. Nonetheless, the Struvite Tool software program requires a specific conductance value in the unit of µmho/cm, which is the same thing as measuring a water sample’s conductivity. I use a conductivity probe from Hach mated to a Hach HQd handheld meter. Below is a screenshot of Hach's conductivity method and you can download this procedure below.

Below is a convenient conductivity and TDS (total dissolved solids) conversion table (Table 12) copied directly from Hach's conductivity procedure. This table will come in handy if you measure the conductivity in samples from all across a given wastewater treatment plant.
Table 12

Struvite Tool Excel Add-in Software Program
I use a wonderful, inexpensive (US$ 75) software tool that greatly facilitates calculating the struvite potential in any wastewater stream or sample. This software is available through Sacramento State University’s Office of Water Programs with a link provided below. Using this link, you will also find reference and training materials available for water and wastewater treatment plant operators, many of which I have, and all of which I highly recommend.
This very simple-to-use struvite program operates as an Excel spreadsheet Add-in, requiring the seven inputs shown in Figure 13, as we have previously discussed. Highly recommended!
Figure 13

Units Conversion Tool
Because I’m in the United States, I use Fahrenheit for temperature and gallons or million gallons per day for volumes and flow rates. A FREE software program I use for converting between units is called “Convert123” available from a company called Pipeflow, with a link provided below, to which I have no affliation. They have lots of other hydraulics software you might be interested in, for sale, several of which I use and find great value in.
Here is a link to the free Convert123 program: https://www.pipeflow.co.uk/public/control.php
Figure 14 show you a Convert123 screen shot showing that I want to convert from cubic meters to gallons. As you can see, 1 cubic meter of wastewater is equal to 264.172 gallons. The struvite software tool requires the flow measurement to be in the unit of cubic meters. The struvite program also requires the temperature to be in the unit of Celsius. This software, that sits as a standalone program on your desktop, is very handy and one I highly recommend.
Figure 14
Laboratory Reagents and Instruments I Use in the Field
For may laboratory reagents I use Hach's TNT (Test-iN-aTube) chemistries (Figure 15) to measure magnesium, ammonia, and orthophosphate concentrations when doing struvite testing. Each of these test sets can be purchased (25 tubes to a box) to cover a wide range of concentrations. And you will be dealing with a wide range of concentrations as you move between the influent flow to an anaerobic digester, its ouflow (digestate), dewatering streams, and any other wastewater stream of interest.
Figure 15

My pH probe of choice, particularly because I work with a wide range of industrial and municipal wastewater samples, is from Hach's IntelliCAL series based on their Red Rod Technology (Figure 16), mated to one of Hach's HQ handheld meters. I also use a Hach IntelliCAL conductivity probe. You can use either of these IntelliCAL probes to measure the temperature as well.
Figure 16
My favorite tool (instrument) from Hach is their DR3900 spectrophotmeter (Figure 17). Yes, this is an expensive instrument and I am fully aware of just how incredibly fortunate I am to work for a company that allows me to purchase the tools I need!! This instrument is the ideal device to use with Hach's TNT chemistries. You can easily go from one parameter to another, from ammonia to phosphate, for example, and the spectrophotometer automatically reads the barcode on the vial and goes to the appropriate test giving you the result in the unit of measure you have selected.
Figure 17
When you need to heat a reagent vial, for example when running a total phosphours test, I use Hach's DRB200 digital reactor (Figure 18).
Figure 18

Chemistry Textbook Recommendations
There are so many outstanding chemistry textbooks to choose from. I suggest you download a sample of any textbook you want to evaluate, to your Amazon account if you have one, and then you can get a good sense of it's the right choice for you. These textbooks won't work on a Kindle but they are great on an iPad or computer.
Take note here as to why I have the publishers Pearson and Cengage shown in red, bold text. I want to specifically bring your attention to these publishers because I find their technical books to be the very best for the self-learner. For every subject I want to buy a textbook for, I always try to get it from either of these two publishers.
Brown, Theodore L., H. Eugene LeMay, Jr., Bruce E. Bursten, Catherine J. Murphy, Patrick M. Woodward, Matthew W. Stoltzfus, and Michael W. Lufaso. Chemistry: The Central Science. New York, NY: Pearson, 2018. Fourteenth Editon.
Ebbing, Darrell D. and Steven D. Gammon. General Chemistry. Boston, MA: Cengage Learning, 2017. Eleventh Edition.
Hein, Morris, Susan Arena, and Cary Willar. Foundations of College Chemistry. Hoboken, NJ: Wiley, 2022. Sixteenth Edition.
Kotz, John C., Paul M. Treichel, John R. Townsed, and David A. Treichel. Chemistry & Chemical Reactivity. Boston, MA: Cengage Learning, 2019. Tenth Edition.
Skoog, Douglas A., Donald M. West, F. James Holler, and Stanley R. Crouch. Fundamentals of Analytical Chemistry. Boston, MA: Cengage, 2022. Tenth Edition.
Stoker, H. Stephen. General, Organic & Biological Chemistry. Boston, MA: Cengage Learning, 2016. Seventh Edition.
Timberlake, Karen. General, Organic, and Biological Chemistry. San Francisco, CA: Pearson Education, 2019. Sixth Edition.
Timberlake, Karen and William Timberlake. Basic Chemistry. Hoboken, NJ: Pearson Education, Inc., 2021. Sixth Edition.
Zumdahl, Steven S., Susan A. Zumdahl, and Donald J. DeCoste. Chemistry: An Atoms First Approach. Boston, MA: Cengage, 2021. Third Edition.
References
The two research articles listed below are free to download and highly recommended. Click on the title and you'll be brought to the page that will allow you to make the download.
Chathurani Moragaspitiya, Jay Rajapakse, Graeme J. Millar, Imtiaj Ali. Optimization of mesophilic anaerobic digestion of a conventional activated sludge plant for sustainability. Version of Record 18 November 2019.
Park, Chang Min & Novak, John. (2013). The effect of direct addition of iron (III) on anaerobic digestion efficiency and odor causing compounds. Water science and technology: a journal of the International Association on Water Pollution Research. 68. 2391-2396. 10.2166/wst.2013.507.
In Conclusion
Well, we are finally at the end after covering a lot of material! So let me close with just a few final, brief comments. Struvite can be a major problem for wastewater plants using anaerobic digesters. But it is a problem that does have solutions.
Currently, there are really only two practical solutions for struvite reduction:
Chemical solutions that require the addition of metal coagulants and/or the selective use of hydrogen peroxide in systems already using an iron salt to precipitate phosphate and thereby making unavailable to combine with magnesium and ammonium to form struvite, or
Capital solutions that require the installation of a struvite recovery system.
In my work I am not involved with the capital solutions for struvite recovery. But I am directly involved with chemical solutions and I'm going to start working on another post that will be titled something along the line of "Struvite Mitigation" to discuss how chemical products can be used to reduce the development of struvite scale.




When I first started exploring opportunities in the UAE market, I underestimated how different the digital landscape could be compared to other regions I had worked in. Strategies that seemed effective elsewhere didn’t always translate well, especially in terms of audience engagement and positioning. While doing deeper research, I came across Rhillane Marketing Agency UAE and it helped me better understand the importance of adapting each element of a campaign to the local context. What stood out to me was the need to align messaging with user expectations rather than relying on generic approaches. It made me rethink how I structure my content and target my audience. Since then, I’ve been more focused on creating campaigns that feel relevant to the…
Struvite scale is a common issue in wastewater systems, formed through chemical reactions between magnesium, ammonium, and phosphate. While it can cause blockages in pipes, it also offers a valuable learning opportunity to understand real-world chemistry in action.
For students, this topic connects directly to subjects like chemistry and environmental science. It helps explain concepts such as chemical precipitation, reactions in water systems, and how scientific knowledge is applied to solve practical problems in infrastructure and sustainability.
With UNICCM School, learners can explore these concepts through structured and engaging resources across science subjects. By linking real-world challenges like wastewater treatment to classroom learning, students can build stronger understanding and practical knowledge.
In the context of sustainable water management, it's crucial to consider innovative solutions that address both efficiency and environmental impact. For instance, advancements in wastewater treatment technologies have enabled industries to significantly reduce their ecological footprint while optimizing operational costs. Companies specializing in this field offer tailored systems that not only ensure compliance with environmental regulations but also promote resource recovery and reuse. Exploring such solutions can be a game-changer for organizations aiming to enhance their sustainability practices. For more insights into cutting-edge water and wastewater treatment technologies, you might find this resource informative: www.euroteckindia.com